ERA®検査 (子宮内膜着床能検査)とは?
- ERA®検査は、Igenomix社が開発し特許を取得した(PCT/ ES2009/000386) (PCT/ ES2009/000386)、子宮内膜着床能を評価する ための検査です。
- ERA®検査は、患者様ごとに胚移植に適したタイミングを見極める初の検査です。
これにより 胚移植を着床のタイミングで合わせて行うことができます。

検査の流れ
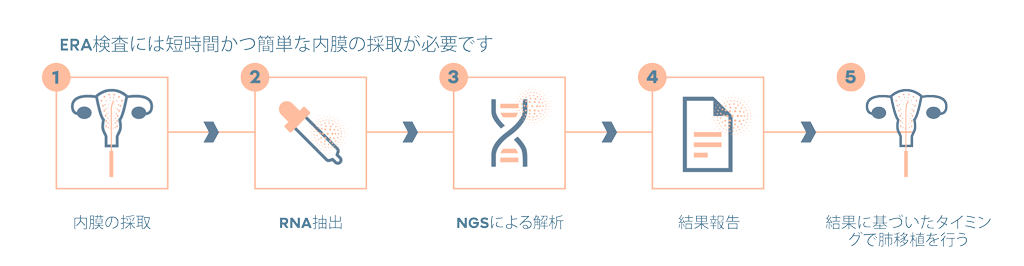
良質な胚を無駄にすることなく、妊娠の可能性を高めます。




検査の90 %は再生検が不要です。再生検は次の2つの場合に行います:

再検査は5%未満です
採取した子宮内膜が検査に十分な量・質でなかった場合、結果が得られず再生検が必要となる場合があります(5%未満)。

必ず妊娠するわけではありません
着床は妊娠を保証するものではありません。胚の質が高くない、あるいは遺伝子の異常や既往症により、妊娠の可能性は低くなる場合があります。

子宮内の状態を評価・予測するものではありません
ERA®検査は他の子宮内膜病変を検査するものではなく、妊娠状態を評価したり、あるいは予測したりするものではありません。
*Simón et al., In vitro fertilization with personalized blastocyst transfer versus frozen or fresh blastocyst transfer: a multicenter, randomized clinical trial. Fertility and Sterility, 2019; 112. e56-e57. 10.1016/j.fertnstert.2019.07.273.
** Ruiz-Alonso et al., The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure. Fertility and Sterility, 2013;100(3):818-24. 10.1016/j.fertnstert.2013.05.004.
A genomic diagnostic tool for human endometrial receptivity based on the transcriptomic signature.
Díaz-Gimeno P, Horcajadas JA, Martínez-Conejero JA, Esteban FJ, Alamá P, Pellicer A, Simón C.
Fertil Steril. 2011 Jan;95(1):50-60, 60.e1-15. doi: 10.1016/j.fertnstert.2010.04.063. Epub 2010 Jul 8.
PMID:20619403
Díaz-Gimeno P, Ruiz-Alonso M, Blesa D, Bosch N, Martínez-Conejero JA, Alamá P, Garrido N, Pellicer A, Simón C.
Fertil Steril. 2013 Feb;99(2):508-17. doi: 10.1016/j.fertnstert.2012.09.046. Epub 2012 Oct 23.
PMID:23102856
Ruiz-Alonso M, Blesa D, Díaz-Gimeno P, Gómez E, Fernández-Sánchez M, Carranza F, Carrera J, Vilella F, Pellicer A, Simón C.
Fertil Steril. 2013 Sep;100(3):818-24. doi: 10.1016/j.fertnstert.2013.05.004. Epub 2013 Jun 4.
PMID:23756099
Bermejo A, Iglesias C, Ruiz-Alonso M, Blesa D, Simón C, Pellicer A, García-Velasco J.
Hum Reprod. 2014 Jun;29(6):1271-8. doi: 10.1093/humrep/deu065. Epub 2014 Apr 4.
PMID:24706003
Ruiz-Alonso M, Galindo N, Pellicer A, Simón C.
Hum Reprod. 2014 Jun;29(6):1244-7. doi: 10.1093/humrep/deu070. Epub 2014 Apr 15.
PMID:24737781
Nejat EJ, Ruiz-Alonso M, Simón C, Meier UT.
Fertil Steril. 2014 Nov;102(5):1477-81. doi: 10.1016/j.fertnstert.2014.07.1254. Epub 2014 Sep 17.
PMID25241377
Bermejo A, Cerrillo M, Ruiz-Alonso M, Blesa D, Simón C, Pellicer A, Garcia-Velasco JA.
Fertil Steril. 2014 Jan;101(1):138-146.e3. doi: 10.1016/j.fertnstert.2013.09.033. Epub 2013 Oct 29.
PMID:24182413
Is endometrial receptivity transcriptomics affected in women with endometriosis? A pilot study.
Garcia-Velasco JA, Fassbender A, Ruiz-Alonso M, Blesa D, D’Hooghe T, Simon C.
Reprod Biomed Online. 2015 Nov;31(5):647-54. doi: 10.1016/j.rbmo.2015.07.014. Epub 2015 Aug 11.
PMID:26385059
Strug MR, Su R, Young JE, Dodds WG, Shavell VI, Díaz-Gimeno P, Ruíz-Alonso M, Simón C, Lessey BA, Leach RE, Fazleabas AT.
Hum Reprod. 2016 Jul;31(7):1552-61. doi: 10.1093/humrep/dew080. Epub 2016 Apr 26.
PMID:27122490
Comstock IA, Diaz-Gimeno P, Cabanillas S, Bellver J, Sebastian-Leon P, Shah M, Schutt A, Valdes CT, Ruiz-Alonso M, Valbuena D, Simon C, Lathi RB.
Fertil Steril. 2017 Mar;107(3):740-748.e2. doi: 10.1016/j.fertnstert.2016.11.009. Epub 2016 Dec 2.
PMID:27919438
Díaz-Gimeno P, Ruiz-Alonso M, Sebastian-Leon P, Pellicer A, Valbuena D, Simón C.
Fertil Steril. 2017 Oct;108(4):703-710.e3. doi: 10.1016/j.fertnstert.2017.07.007. Epub 2017 Aug 30.
PMID:28863933
Inter-cycle consistency versus test compliance in endometrial receptivity analysis test.
Stankewicz T, Valbuena D, Ruiz-Alonso M.
J Assist Reprod Genet. 2018 May 26. doi: 10.1007/s10815-018-1212-7
Effect of mifepristone on the transcriptomic signature of endometrial receptivity.
von Grothusen C, Lalitkumar PG, Ruiz-Alonso M, Boggavarapu NR, Navarro R, Miravet-Valenciano J, Gemzell-Danielsson K, Simon C.
Hum Reprod. 2018 Aug 22. doi: 10.1093/humrep/dey272. [Epub ahead of print] PMID: 30137464
Live birth after embryo transfer in an unresponsive thin endometrium.
Gynecol Endocrinol. 2014 Jul;30(7):481-4. doi: 10.3109/09513590.2014.900747. Epub 2014 Mar 20.
Endometrial receptivity array: Clinical application.
Mahajan N.
J Hum Reprod Sci. 2015 Jul-Sep;8(3):121-9. doi: 10.4103/0974-1208.165153. Review.
PMID:26538853
Hashimoto T, Koizumi M, Doshida M, Toya M, Sagara E, Oka N, Nakajo Y, Aono N, Igarashi H, Kyono K.
Reprod Med Biol. 2017 Jun 27;16(3):290-296. doi: 10.1002/rmb2.12041. eCollection 2017 Jul.
PMID:29259480
Tan J, Kan A, Hitkari J, Taylor B, Tallon N, Warraich G, Yuzpe A, Nakhuda G.
J Assist Reprod Genet. 2018 Jan 11. doi: 10.1007/s10815-017-1112-2.
PMID:29327111
The Reproductive Outcomes for the Infertile Patients with Recurrent Implantation Failures May Be Improved by Endometrial Receptivity Array Test.
Ota, T., Funabiki, M., Tada, Y., Karita, M., Hayashi, T., Maeda, K. et al.
Journal of Medical Cases. 2019; 10(5), 138-140. doi: https://doi.org/10.14740/jmc3282
Why results of endometrial receptivity assay testing should not be discounted in recurrent implantation failure?
Simrandeep K., Padmaja N. The Onco Fertility Journal. 2019; 2(1): 46-49.
Personalized Embryo Transfer Helps in Improving In vitro Fertilization/ICSI Outcomes in Patients with Recurrent Implantation Failure.
Patel JA, Patel AJ, Banker JM, Shah SI, Banker MR. J Hum Reprod Sci. 2019; 12(1):59-66. doi: 10.4103/jhrs.JHRS_74_18.
Endometrial Receptivity Analysis – a tool to increase an implantation rate in assisted reproduction.
Hromadová L; Tokareva I; Veselá K; Trávník P; Veselý J. Ceska Gynekol. 2019; 84(3): 177-183.
Barrenetxea G, Romero I, Celis R, Abio A, Bilbao M, Barrenetxea J. Eur J Obstet Gynecol Reprod Biol. 2021 Aug;263:192-197.